Stakeholders have four weeks to respond
EC launches EU health claims ‘fitness check’; nutrient profiles and botanicals targeted

Stakeholders have four weeks to give input into the roadmap which avoids a broader assessment of the controversial regulation “given that the list of authorised health claims only came into application in December 2012.” Such a roadmap may come later.
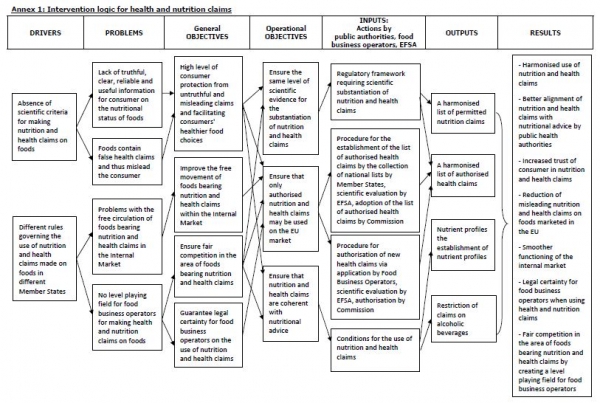
Eight years after the EU nutrition and health claims regulation (NHCR) was written into law the EC wants to discover whether it is “fit for purpose” and achieved “its overall objectives on truthful information to consumers and the facilitation of the free movement of foods bearing claims.”
It would seek to determine whether much-debated nutrient profile models "warranted and adequate to ensure the objectives of the Regulation".
Five criteria will drive the assessment:
- Effectiveness
- Efficiency
- Relevance
- Coherence
- EU added value
Botanicals under the microscope
The European Federation of Associations of Health Product Manufacturers (EHPM) welcomed the EC move, especially in regard to the EU’s €2bn botanicals market (of €9bn for food supplements in total).
Under the NHCR there are about 1500 botanical health claim submission on-hold since 2011. Before then about 500 were rejected by the European Food Safety Authority (EFSA).
“EHPM thinks that this is a very positive development and we hope it can lead to the development of a workable regulatory structure for botanicals,” said director general Patrick Ahern, noting the group had already sent its policy priorities to the EC.
These included an acknowledgement nutrient-based science should not be treated in the same way as pharma science. “Foods are not drugs and should not be assessed as if they are,” EHPM said in that statement.
"Numerous health claims have been dismissed by EFSA which were previously approved by national authorities when this area was regulated at national level."
It added: "EHPM urges the Commission to respect national traditions and best practice when formulating an appropriate policy."
The EC roadmap preamble acknowledges that in the case of botanicals there has been much debate over the usability of ‘tradition of use’ data in backing health claims, and noting how such data has been more readily admissible under the Traditional Herbal Medicinal Products Directive (THMPD), which since 2011 has offered a route to market for herbal products in the EU that lies between food and medicines laws.
“Under the legislation on medicinal products for human use, herbal medicinal products may undergo a simplified registration procedure instead of an authorisation procedure on the basis of criteria specified in the legislation on medicinal products for human use such as evidence on medicinal use throughout a period of at least 30 years 'traditional use',” the EC report states.